- English
- French
- German
- Portuguese
- Spanish
- Russian
- Japanese
- Korean
- Arabic
- Greek
- German
- Turkish
- Italian
- Danish
- Romanian
- Indonesian
- Czech
- Afrikaans
- Swedish
- Polish
- Basque
- Catalan
- Esperanto
- Hindi
- Lao
- Albanian
- Amharic
- Armenian
- Azerbaijani
- Belarusian
- Bengali
- Bosnian
- Bulgarian
- Cebuano
- Chichewa
- Corsican
- Croatian
- Dutch
- Estonian
- Filipino
- Finnish
- Frisian
- Galician
- Georgian
- Gujarati
- Haitian
- Hausa
- Hawaiian
- Hebrew
- Hmong
- Hungarian
- Icelandic
- Igbo
- Javanese
- Kannada
- Kazakh
- Khmer
- Kurdish
- Kyrgyz
- Latin
- Latvian
- Lithuanian
- Luxembou..
- Macedonian
- Malagasy
- Malay
- Malayalam
- Maltese
- Maori
- Marathi
- Mongolian
- Burmese
- Nepali
- Norwegian
- Pashto
- Persian
- Punjabi
- Serbian
- Sesotho
- Sinhala
- Slovak
- Slovenian
- Somali
- Samoan
- Scots Gaelic
- Shona
- Sindhi
- Sundanese
- Swahili
- Tajik
- Tamil
- Telugu
- Thai
- Ukrainian
- Urdu
- Uzbek
- Vietnamese
- Welsh
- Xhosa
- Yiddish
- Yoruba
- Zulu
Are Custom LCD Display Modules Suitable for Medical Devices?
Custom LCD display modules represent a compelling solution for medical device applications, delivering tailored visual interfaces that meet healthcare's stringent requirements. Unlike generic off-the-shelf screens, these specialized modules offer adaptable sizes, enhanced clarity, and interfaces designed specifically for medical-grade performance. They offer dependable performance in sterilization processes, temperature swings, and demanding clinical settings when carefully integrated. Choosing the right modules is crucial and should be based on their certifications, strong build quality, and ability to fit well with your device's design and regulatory requirements.

Understanding Custom LCD Display Modules in Medical Devices
Medical devices demand display solutions that exceed consumer electronics standards. When we talk about LCD display modules designed for healthcare, we're discussing components engineered to withstand rigorous sterilization cycles, maintain accuracy under varying temperatures, and deliver consistent performance across extended operational lifespans.
Core Technologies Behind Medical-Grade Displays
Display material biocompatibility matters in direct or indirect patient touch. ISO 10993 testing ensures materials won't harm biology. RoHS and REACH compliance is crucial for electronics sold in Europe, where regulations are severe.
Any reliable medical display relies on its technology. TN (Twisted Nematic) panels have quick response times for real-time monitoring equipment but limited viewing angles. For diagnostic imaging, where many doctors assess results, IPS (In-Plane Switching) technology provides greater color fidelity and wide viewing angles. For precise patient data visualization, TFT displays with active matrix capabilities offer higher resolutions and contrast ratios.
Specific backlight units for long-term operation are used in these modules. Displays for medical infusion pumps and ventilators must remain visible for 50,000+ hours without brightness loss. Understanding these basic technologies helps procurement teams choose a device-specific display architecture.
Interface Types and Integration Considerations
Communication protocols significantly impact development complexity and system performance. SPI (Serial Peripheral Interface) offers straightforward implementation with minimal pin requirements, making it ideal for battery-powered portable devices where simplicity matters. LVDS (Low-Voltage Differential Signaling) supports the higher data throughput necessary for larger diagnostic screens displaying complex waveforms or imaging data. I2C interfaces allow multiple devices to communicate with each other, which is helpful in medical systems where different parts need to share information
Selecting the appropriate interface requires balancing data transmission speed, power consumption, and your existing hardware architecture. A custom LCD module with flexible interface options reduces redesign costs when adapting devices for different market segments or regulatory regions.
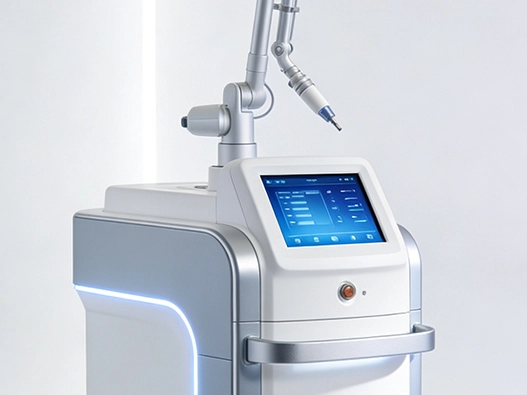
Advantages of Custom LCD Display Modules for Medical Devices
Medical applications present unique challenges that custom display solutions address effectively. The ability to tailor every aspect of the visual interface creates measurable advantages in device performance, regulatory compliance, and user safety.
Superior Visual Accuracy for Diagnostic Precision
Display material biocompatibility matters in direct or indirect patient touch. ISO 10993 testing ensures materials won't harm biology. RoHS and REACH compliance is crucial for electronics sold in Europe, where regulations are severe.
Color reproduction accuracy affects the reliability. Medical LCD display modules with Delta E values below 2 ensure color consistency when doctors evaluate tissue colors, blood oxygenation, and diagnostic imaging data. Resolution adjustment lets you tailor pixel density to the visualization task—vital sign monitors need different specs than surgical navigation systems showing dense anatomical structures.
Display brightness uniformity minimizes medical data misunderstanding. Standard commercial displays often vary in brightness by over 20% between the center and edge. Medical-grade bespoke modules present data uniformly within 5% regardless of viewing position during procedures.
Durability Under Clinical Conditions
Healthcare environments expose equipment to aggressive cleaning protocols. Custom LCD display modules built for medical applications feature sealed constructions with IP65 or higher ingress protection, preventing liquid penetration during repeated disinfection with alcohol, bleach, or hydrogen peroxide solutions. The bonding technology between display layers—whether air gap or optical bonding—affects both durability and optical clarity under chemical exposure.
Temperature resilience ensures reliable operation across diverse clinical settings. Operating rooms maintain cooler temperatures, while equipment stored in ambulances experiences extreme heat. Custom modules engineered for -20°C to +70°C operational ranges maintain performance consistency across these environments, preventing display failures that could compromise patient care.

Flexible Integration with Medical Device Architectures
Display material biocompatibility matters in direct or indirect patient touch. ISO 10993 testing ensures materials won't harm biology. RoHS and REACH compliance is crucial for electronics sold in Europe, where regulations are severe.
Modern medical gadgets have power management, data processing, wireless networking, and user interface subsystems. Custom display modules with flexible mounting, connection, and electrical characteristics ease device integration. This flexibility speeds development, decreasing medical device manufacturers' time-to-market pressures.
The GUITION ESP32-8048S070N shows this integration advantage. This 7-inch display module, powered by the ESP32-S3-WROOM-1 dual-core MCU at 240 MHz, features 800x480 resolution and Wi-Fi and Bluetooth connections. The 512KB SRAM, 8M PSRAM, and 16MB flash provide ample processing and storage for complicated medical interfaces. Reserved TF card and IO port interfaces allow device feature expansion during the product lifecycle.
With multi-platform development support, this medical prototype module stands out. Engineering teams can use Arduino IDE, ESP IDE, Micropython, or Mixly to match their skills instead of learning proprietary programming tools. Compatibility shortens the learning curve and development cycle, which is crucial when meeting FDA submission deadlines or responding to evolving clinical demands.
Energy Efficiency for Portable Medical Equipment
Battery life directly impacts the usability of portable diagnostic devices, patient monitors, and wearable medical sensors. Custom LCD display modules optimized for low power consumption extend operational duration between charges. Advanced backlight control circuits adjust brightness based on ambient conditions, reducing unnecessary power draw while maintaining readability.
Dynamic power management techniques dim displays during periods of inactivity without requiring a complete shutdown, preserving battery capacity during extended patient monitoring sessions. These efficiency gains translate to fewer battery replacements, reduced device weight, and improved clinician satisfaction with portable equipment performance.
Key Considerations When Choosing Custom LCD Display Modules for Medical Devices?
Selecting the right display module requires balancing technical specifications, regulatory requirements, and practical procurement considerations. Medical device manufacturers must evaluate multiple factors to ensure chosen components support both device functionality and compliance pathways.
Regulatory Compliance and Certifications
Markets regulate medical devices differently. CE certification under the Medical Device Regulation (MDR) restricts European market access, whereas FDA approval controls US distribution. ISO 13485 accreditation shows a supplier's commitment to managing the quality of medical devices. Checking that your custom LCD display module provider has these certifications decreases device submission compliance concerns.
Display material biocompatibility matters in direct or indirect patient touch. ISO 10993 testing ensures materials won't harm biology. RoHS and REACH compliance is crucial for electronics sold in Europe, where regulations are severe.
Display Resolution and Size Alignment
Display specs should match clinical use cases to avoid overengineering and functional issues. Bedside patient monitors with six vital sign parameters require different resolutions than surgical navigation systems with 3D anatomical models. Data density, viewing distance, and clinical workflow should be carefully considered to avoid picking displays that don't match visualization objectives or increase device expenses.
Device portability and clinical workflow integration of the custom LCD display module depend on screen size. Compact 3.5-inch to 5-inch displays balance information density and device weight for handheld diagnostic equipment. Clinically active doctors may use stationary devices like infusion pump controllers with 7-inch to 10-inch screens for larger touch targets and better reading.
Interface Selection Impact on System Performance
The communication protocol between your main controller and display module affects refresh rates, power consumption, and development complexity. SPI interfaces simplify firmware development but may limit frame rates on larger displays with high resolutions. LVDS supports the bandwidth requirements of diagnostic imaging displays where smooth, real-time video is essential.
Evaluating your processor's available communication channels helps narrow interface choices. The ESP32-S3 platform in the GUITION module provides multiple interface options, offering flexibility during prototype development when system architecture may still evolve based on testing feedback.
Supplier Reliability and Long-Term Availability
Medical devices often remain in production for 10-15 years, yet display component lifecycles typically span 3-5 years in consumer markets. Establishing relationships with custom LCD module suppliers who commit to extended component availability prevents costly redesigns when displays reach end-of-life. Suppliers offering product lifecycle management services provide advance notice of component changes, enabling proactive qualification of replacement parts.
Quality control processes at the supplier level directly impact your device reliability. Suppliers employing automated optical inspection, environmental stress screening, and comprehensive functional testing deliver more consistent component quality, reducing field failure rates and associated warranty costs.
Implementing Custom LCD Display Modules in Medical Devices: Design and Procurement Insights
Successful integration of display modules requires coordinated efforts across engineering, regulatory, and supply chain functions. Learning from established implementation patterns helps teams avoid common pitfalls while accelerating development timelines.
Design Principles for Medical User Interfaces
Clinicians' use of time-pressure medical gadgets is affected by ergonomics. Touch targets should be larger than 9mm x 9mm for gloved operation in sterile settings. Color coding for colorblind people requires additional visual cues. Clinical settings have different lighting conditions; therefore, font sizes must be legible.
Additional sterilization is possible beyond the display module. Bezels, mounting hardware, and external cabling must last many cleanings. Optical bonding eliminates air gaps between display layers, generating smooth surfaces that resist bacterial colonization and improve contrast ratios under bright ambient light.
Real-World Implementation Examples
Custom displays work in patient monitoring systems for intensive care units. Twelve- to fifteen-inch high-brightness displays ensure vision under surgical lights. To avoid visual clutter, multi-parameter displays must have good information architecture and enough resolution to show ECG waveforms, SpO₂ trends, blood pressure data, and warning messages.
Custom LCD modules are used in battery-powered ultrasound systems. These displays use adaptive lighting control and efficient pixel designs to balance image quality and power consumption. In emergency departments and field medical operations, rugged construction allows point-of-care imaging.
Touchscreen responsiveness and visual clarity are crucial in infusion pump interfaces. Clinicians must program flow rates and drug concentrations accurately for medication distribution. Touchscreens tuned for accurate input and high-contrast displays with intuitive layouts eliminate programming errors.
Procurement Strategies for Medical Device Manufacturing
Establishing clear specifications prevents misunderstandings between device manufacturers and display suppliers. Detailed drawings documenting mechanical dimensions, connector types, pinout configurations, and optical characteristics create shared expectations. Environmental specifications should include operating temperature ranges, humidity tolerance, vibration resistance, and EMI/EMC requirements specific to medical equipment standards.
Minimum order quantities vary significantly between suppliers. Some custom LCD display module manufacturers require 1,000+ unit initial orders, creating cash flow challenges for startups developing novel devices. Identifying suppliers offering lower MOQs or prototyping services enables iterative development without excessive inventory commitments.
Lead time management prevents production delays. Standard displays may ship within 2-4 weeks, while fully customized modules requiring new tooling can extend to 12-16 weeks. Building display selection decisions into early development phases ensures component availability aligns with production ramp schedules.
Why does partnering with experienced custom LCD display module suppliers matter?
The supplier relationship extends far beyond transactional component purchases. Strategic partnerships with display manufacturers create competitive advantages through technical collaboration, supply chain reliability, and ongoing innovation access.
Quality Assurance and Medical Industry Expertise
Medical suppliers know regulatory and testing standards that consumer electronics companies may miss. They maintain design history files and traceability documentation for device submissions and apply FDA 21 CFR Part 820 design controls. Regulatory knowledge reduces component qualifying back-and-forth and speeds approval.
Medical component manufacturing controls differ greatly from those of consumer electronics. Before device manufacturers receive components, material inspection, environmental monitoring, and functional testing detect flaws. Medical applications require constant performance, and statistical process control detects trends before they impair product quality.
Customization Capabilities and Time-to-Market
Experienced suppliers' engineering supports display selection and integration. Based on medical device project experience, their technical teams can provide interface kinds, resolution alternatives, and integration solutions. This advice avoids costly design iterations and development rework.
OEM customization goes beyond specs. Suppliers can alter firmware for specialized communication protocols, adjust lighting control algorithms for clinical conditions, or add ambient light sensors. These customized changes differentiate device functionality without in-house display engineering.
Long-Term Partnership Benefits
Device commercialization makes a consistent component supply more valuable. Medical suppliers avoid allocation concerns during shortages of consumer electronics components by maintaining reliable supply chains. Long-term agreements shield device manufacturers from market instability with pricing stability provisions.
After-sales technical assistance covers production scaling and field deployment difficulties. Suppliers respond to integration issues, supply replacement parts for failure investigation, and manage engineering change management for component changes. This constant partnership ensures device dependability and customer satisfaction across product lifecycles.
Guition shows this cooperation approach through extensive development support. Our engineers supply Arduino library functionalities and comprehensive test programs, decreasing development time. Faster medical device prototype proof-of-concept development is possible with the factory-programmed test software for the ESP32-8048S070N module. Instead of learning proprietary technologies, our multi-platform compatibility lets your staff develop in familiar contexts.

Conclusion
Custom LCD display modules deliver measurable advantages for medical device applications when selected thoughtfully and integrated properly. Their ability to withstand clinical environments, meet regulatory requirements, and provide tailored functionality makes them suitable for diverse healthcare applications ranging from patient monitoring to diagnostic imaging. Success requires careful evaluation of technical specifications, regulatory compliance, and supplier capabilities. The flexibility of modern display modules like the GUITION ESP32-8048S070N, combined with robust development support and multi-platform compatibility, enables medical device manufacturers to accelerate time-to-market while maintaining the quality standards healthcare demands.
FAQ
How do custom LCD modules differ from standard displays in medical reliability?
Custom LCD display modules designed for medical applications undergo enhanced environmental testing, including extended temperature cycling, humidity resistance validation, and chemical compatibility verification. Standard commercial displays typically qualify for consumer environment conditions, lacking the durability testing and documentation required for medical device integration. Medical-grade modules also feature longer operational lifespans, often rated for 50,000+ hours of continuous operation compared to 30,000 hours for consumer displays.
What certifications should we verify when sourcing medical display modules?
Verify that suppliers maintain ISO 13485 certification, demonstrating quality management systems specific to medical devices. Component-level RoHS and REACH compliance ensures hazardous substance restrictions are met. Depending on your device classification and intended markets, you may require displays with biocompatibility testing per ISO 10993 standards. CE and FDA compliance ultimately rests with the device manufacturer, but supplier documentation significantly supports regulatory submissions.
What typical lifespan can we expect from displays in medical devices?
Properly specified custom LCD display modules in medical applications typically achieve a 7-10 year operational life under normal clinical usage patterns. Backlight degradation represents the primary life-limiting factor, with LED backlights gradually dimming over time. Operating displays at reduced brightness levels significantly extends lifespan. Temperature management also impacts longevity—displays operating continuously at upper temperature limits experience accelerated aging compared to those in controlled environments.
Partner with Guition for Your Medical Display Development Needs
Medical device development requires display solutions that balance regulatory compliance, technical performance, and development efficiency. Guition brings specialized expertise in custom LCD display modules designed for demanding applications across healthcare and industrial sectors. Our ESP32-8048S070N module combines powerful ESP32-S3 processing, integrated Wi-Fi and Bluetooth connectivity, and flexible development platform support—creating a foundation for rapid medical device prototyping and production.
As an experienced custom LCD display module supplier, we understand the unique challenges medical device manufacturers face. Our engineering team provides comprehensive technical documentation, responsive support, and customization services that align with your device requirements and timeline pressures. We maintain rigorous quality control processes and long-term component availability commitments that protect your product lifecycle.
Ready to explore how Guiton's display solutions can accelerate your medical device development? Contact our team at david@guition.com to discuss your specific requirements, request technical specifications, or arrange a sample evaluation. We're committed to becoming your trusted partner in creating reliable, compliant, and innovative medical devices.
References
1. International Electrotechnical Commission. Medical electrical equipment - Part 1-2: General requirements for basic safety and essential performance - Collateral standard: Electromagnetic disturbances - Requirements and tests. IEC 60601-1-2:2014.
2. Association for the Advancement of Medical Instrumentation. Human factors engineering—design of medical devices. ANSI/AAMI HE75:2009/(R)2018.
3. U.S. Food and Drug Administration. Design Considerations for Devices Intended for Home Use - Guidance for Industry and Food and Drug Administration Staff. December 2014.
4. International Organization for Standardization. Medical devices - Application of risk management to medical devices. ISO 14971:2019.
5. Society for Information Display. Information Display Standards and Guidelines for Medical Imaging Applications. Journal of the Society for Information Display, Volume 26, Issue 8, 2018.
6. European Commission. Medical Device Coordination Group Document: Guidance on Classification Rules for in vitro Diagnostic Medical Devices under Regulation (EU) 2017/746. MDCG 2020-16, November 2020.
Learn about our latest products and discounts through SMS or email








